What are Gold Nano-particles
These are particles that are created at the nano scale and are formed in a solution. Whether that solution is a deep eutectic solvent or an ionic liquid a reduction reaction happens that turns gold into a deionized gold and allows them to grow into nano-particles.
Deep eutectic solvent
With research moving towards DES rather then IL the first thing was understanding the use and application of DES to AuNPs. DES has advantages over IL as they are easy to make and the components are easily attainable and inexpensive to get. DESs also aren't flammable, exhibit low vapor pressure, and have a wide liquid range3. This allows them to be used in many different types of synthesis with different metals. The use of DES in this experiment would be a reaction medium and a shape-directing agent for the AuNPs. The DESs being cheap also allows them to be used in industrial purposes.
Reline
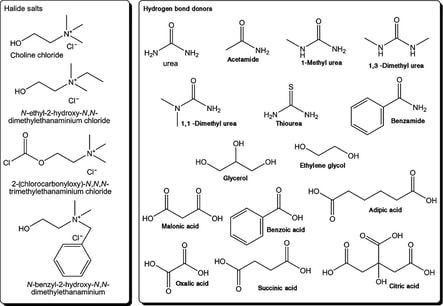
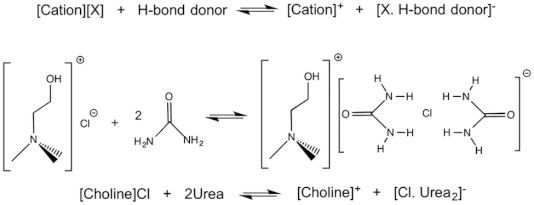
One DES that is used is called reline and is made from chlorine chloride and urea in a 1:2 molar ratio. This is because the chlorine chloride has two places that the urea will create a hydrogen bond due to the valence electrons on the Chlorine and so it will make a structure that can control the shape of the Au NPs.
Abstract
One of the big problems facing the gold nanoparticle industry is that size is hard to control. However size is important when it comes to gold nanoparticles as size is part of what makes them useful. Deep Eutectic Solvents (DES) are an emerging solvent for the synthesis of the gold nanoparticles as DES act as a cheaper and easier to make stabilizer for the shape of the nanoparticles. The DES that was used was reline, a molecule made of chlorine chloride and urea in a 1:2 molar ratio. This research was conducted to analyze how increasing the water concentration of reline solutions would affect the size of the gold nanoparticles during synthesis. It was hypothesised that increasing the concentration of water in the reline solution will increase the average size of the gold nanoparticles because the lack of the stabilizer in the synthesis solution would allow the gold to aggregate into larger nanoparticles. The idea being that with more water there is less of the stabilizing agent in the solution and so the particles will grow bigger. The results of the four trials that were done of the water reline solutions were gathered from a coulometric titrator, UV-Vis, and AFM. The different concentrations of the water used in testing was 2.5mL of water and 7.5mL of reline, 5mL of water and 5mL of reline, 7.5mL of water and 2.5mL of reline, and 10mL of water and 0mL of reline. In the end the results showed that gold nanowires may have been created or nanospheres could have been created. The nanoparticles that were created aggregate and this was seen in the AFM. During synthesis of the 7.5mL of water and 2.5mL of reline the gold aggregated into a macro form leaving none in solution which can be seen in the UV-Vis. With this evidence the hypothesis can be minimally supported but further research would need to be done to better support the hypothesis.